Self adhesive labels are used across many industries, including food packaging, beverage bottles, cosmetics, logistics, and pharmaceuticals. Although labels may appear simple, producing them involves a precise manufacturing process to ensure printing accuracy, strong adhesion, and compatibility with labeling machines.
This article explains how labels are printed step by step, from artwork preparation to final packaging.
1. Artwork Preparation and Prepress
The first stage of label printing is artwork preparation.
Customers usually provide design files in formats such as:
Before production begins, the prepress team checks the design carefully. This process typically includes:
- verifying color modes (Pantone or CMYK)
- checking image resolution
- confirming barcode clarity
- adjusting small text or fine details
- adding bleed areas and die-cut outlines
Proper prepress preparation ensures that the printed labels match the intended design and reduces the risk of production errors.
2. Printing Plate Production
After artwork approval, the next step is printing plate production.
Different printing technologies require different types of plates.
Flexographic printing plates
Flexographic printing uses photopolymer plates. The design is transferred onto the plate using laser imaging technology. These plates then transfer ink onto the label material during printing.
Offset printing plates
Offset printing uses aluminum plates. The plate separates image areas from non-image areas, allowing ink to transfer accurately to the material.
Plate quality directly affects the sharpness and consistency of the printed labels.
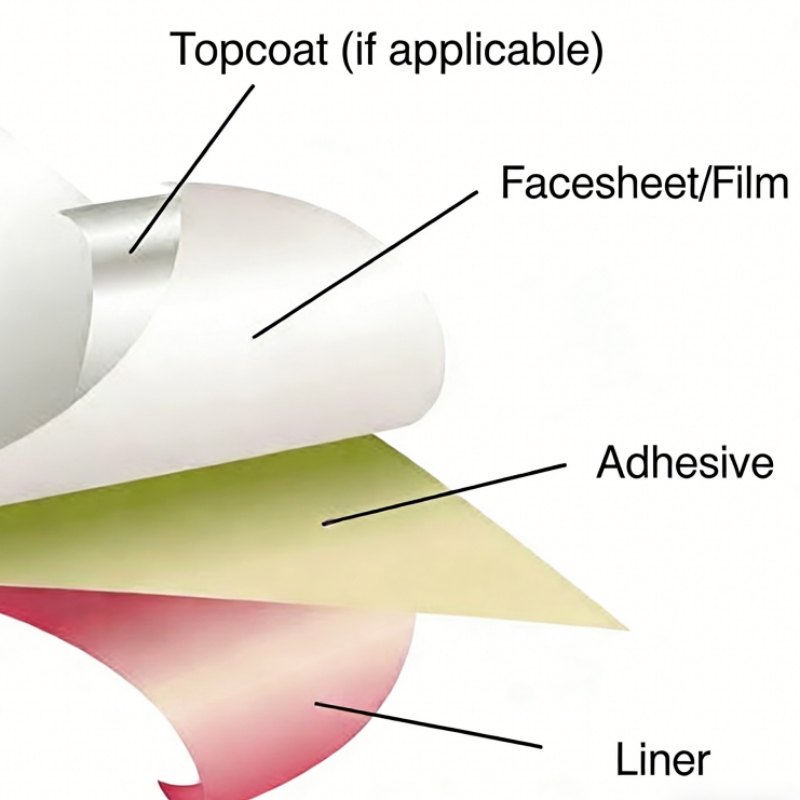
3. Label Material Selection
Before printing begins, manufacturers select the appropriate label material and adhesive according to the application.
Common label materials include:
- paper labels
- BOPP film labels
- PET labels
- PE labels
Each material has different characteristics. For example:
- BOPP labels are often used for beverage bottles because they are waterproof.
- PET labels provide higher durability and chemical resistance.
- Paper labels are commonly used for food and logistics applications.
Adhesives are also selected based on usage conditions, such as:
- permanent adhesive
- removable adhesive
- freezer-grade adhesive
The materials are supplied in large rolls for efficient production.
4. Label Printing Process
The material roll is mounted onto the printing press.
During printing, the following steps occur:
- Ink is transferred to the printing plate
- The plate transfers the design onto the label material
- Multiple printing units apply different colors
Modern label printing machines can print multiple colors in a single pass, ensuring high production efficiency.
Common label printing technologies
Two of the most widely used printing methods are:
Flexographic printing
- suitable for high-speed production
- widely used for packaging labels
- compatible with many materials
Offset printing
- suitable for detailed images and gradients
- excellent color consistency
- often used for high-precision label designs
Both methods are commonly used in professional label manufacturing.
5. Finishing and Surface Treatment
After printing, labels may undergo additional finishing processes to enhance their appearance and durability.
Common finishing options include:
- matte lamination
- gloss lamination
- varnish coating
- hot foil stamping
- spot UV coating
- embossing
These processes can improve both visual appeal and performance, such as resistance to moisture, oil, or abrasion.
6. Die Cutting
Next, the printed material goes through die cutting.
A die-cutting tool cuts the labels into the required shapes and sizes. At the same time, the excess material around the labels is removed.
After die cutting, the labels remain attached to a release liner, making them easy to peel and apply.
Labels can be produced in different formats, including:
- roll labels for automatic labeling machines
- sheet labels for manual use
7. Inspection and Quality Control
After printing and finishing, the labels go through quality inspection before slitting.
Many label manufacturers use automatic inspection systems to detect potential defects across the entire roll.
These systems can identify issues such as:
- color variation
- printing defects
- missing text or graphics
- registration misalignment
Some production lines are equipped with 100% automatic inspection machines, which scan every label on the roll to ensure consistent quality.
Early inspection helps prevent defective labels from continuing to later production stages.
8. Slitting and Rewinding
Once the inspection process is completed, the master roll moves to the slitting machine.
The large roll is cut into smaller rolls according to customer specifications.
During this step, manufacturers control several parameters:
- roll width
- core size
- number of labels per roll
- winding direction
Proper slitting ensures that the finished label rolls can run smoothly on the customer’s labeling machines.
9. Packaging and Shipment
Finally, the finished label rolls are carefully packed for shipment.
Packaging methods may include:
- protective plastic wrapping
- carton packaging
- pallet packaging for large shipments
Proper packaging protects the labels from moisture, dust, and damage during transportation.
Conclusion
Printing self adhesive labels involves several precise production steps, including artwork preparation, plate making, printing, finishing, die cutting, inspection, and slitting.
Each step plays an important role in ensuring that the labels meet quality requirements and perform reliably during application.
With modern technologies such as flexographic and offset printing, label manufacturers can produce customized labels suitable for a wide range of packaging applications.
FAQ
What printing methods are used for labels?
The most common label printing methods include flexographic printing, offset printing, and digital printing. Flexographic printing is widely used for large production volumes.
Why is inspection important in label production?
Inspection helps detect printing defects, color variations, or missing elements. Many manufacturers use automatic inspection systems to ensure consistent quality.
What materials are commonly used for labels?
Common label materials include paper, BOPP film, PET, and PE. The choice depends on the product application and environmental conditions.